ASHP Non Sterile Compounding: Best Practices for Pharmacies and Healthcare Facilities

Non sterile compounding is an important aspect of pharmacy practice, allowing for the customization of medications to meet individual patient needs. By adhering to ASHP non sterile compounding standards, pharmacies can ensure compounded preparations’ safety, accuracy, and effectiveness.
This article explores the definition of non-sterile compounding, the basic requirements of USP 795 guidelines, and how to create a safe environment while compounding non-sterile hazardous drugs.
What Is Non-Sterile Compounding?
It is the process of modifying a drug or bulk drug ingredient to produce a customized preparation for individual patient needs. The process involves admixing, pooling, diluting, and reconstituting pharmaceutical ingredients. Unlike sterile compounding, non sterile compounding doesn’t require a sterile environment.
Non-Sterile Compounds In Healthcare Facilities
Healthcare facilities carefully prepare non-sterile compounds to maintain the purity and potency of the active ingredients. It is also important to ensure precise mixing, packaging, labeling, and storage of the final product while keeping all surfaces, materials, and equipment clean.
Types of non-sterile formulations include:
- Topical gels, ointments, creams, and lotions.
- Powders and suppositories.
- Oral solutions and suspensions.
- Capsules and tablets.
ASHP Non Sterile Compounding Guidelines
The American Society of Health-System Pharmacists (ASHP) is the largest organization of health professional pharmacists in the United States, representing all healthcare settings. Through its extensive resources, it helps pharmacy professionals enhance their skills.
ASHP offers recommendations to guarantee that non sterile compounding procedures in medical institutions are safe and effective. ASHP non sterile compounding guidelines follow the USP 795 chapter.
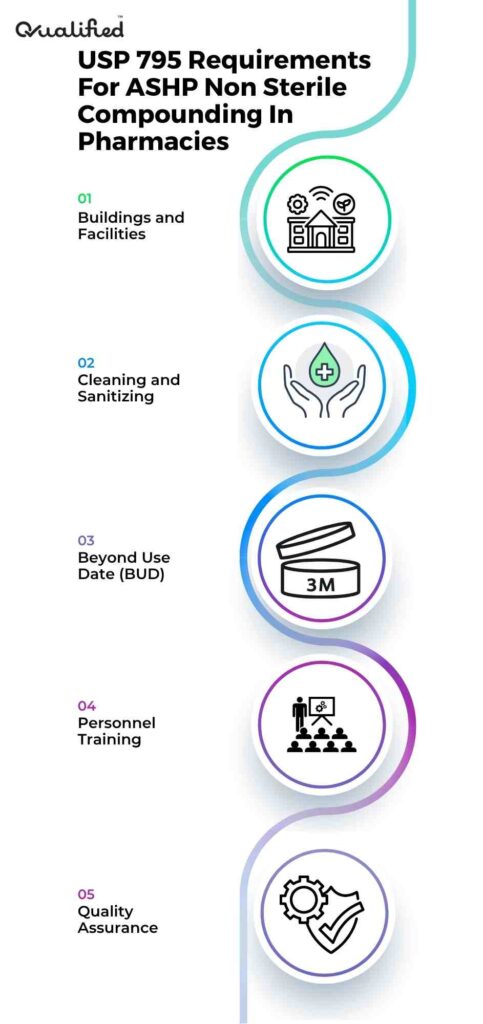
USP 795 Requirements For ASHP Non Sterile Compounding In Pharmacies
USP 795 requirements for non-sterile compounding include the following:
1. Buildings and Facilities
A specific area is required for non sterile compounding, away from other activities. A compounding pharmacist must monitor the temperature daily in storage areas, and proper water sources must be available for cleaning and rinsing.
2. Cleaning and Sanitizing
Non-sterile compounding areas must be cleaned and sanitized regularly, following specific frequencies determined by USP 795. When selecting cleaning and sanitizing agents, it is crucial to consider their compatibility, effectiveness, and potential to leave residues.
3. Beyond Use Date (BUD)
BUD is the period after compounding during which a compounded preparation remains safe and effective. As a qualified pharmacist, you must consider many factors when establishing BUD, such as the active ingredients’ chemical and physical stability, the risk of microbial growth, and the compatibility of the container–closure system to maintain the preparation’s integrity and potency.
4. Personnel Training
All personnel preparing and handling compounded non-sterile preparations (CNSPs) must undergo initial training, including hand hygiene, garbing, cleaning, equipment use, documentation, handling, and transporting components and CNSPs.
5. Quality Assurance
The quality assurance program is important to ensure that CNSPs are prepared in compliance with USP 795 guidelines, providing high-quality compounds. A designated person must oversee the implementation of this program, ensuring the prevention and detection of errors, evaluating adverse events and appropriate corrective actions.
Enhancing your expertise in non-sterile compounding is crucial for ensuring patient safety and maintaining compliance with ASHP non sterile compounding guidelines. Know more about the differences between sterile & non-sterile preparations through our ultimate Sterile Compounding & Aseptic Technique Course
Compounding Non Sterile Hazardous Drugs: A Safe Environment
Compounding non-sterile hazardous drugs requires strict adherence to safety protocols to provide a safe environment for personnel and patients. For procedures like weighing non-sterile powders, facilities must use Containment Primary Engineering Controls (C-PECs), such as Biological Safety Cabinet (BSC), and Class I Containment Ventilated Enclosures (CVEs), per USP 800 standards.
Additionally, a C-PEC intended for sterile hazardous compounding must be completely cleaned, disinfected, and decontaminated before and after each usage to avoid cross contamination when used for non sterile compounding. Thus preserving the integrity of the compounded drugs and guaranteeing a safe environment.
Conclusion
In conclusion, non-sterile compounding is essential for providing customized, safe, and effective medications. By adhering to ASHP non sterile compounding guidelines, pharmacists can ensure the highest safety and efficacy standards while providing personalized, patient-centered care. To achieve this, it is necessary to ensure continuous learning, knowledge of the latest updates, expansion of skills, and keeping up with continuous professional development CPD. Follow our blog for more interesting medical articles.
recent posts :
Top Rated Courses :

Upcoming Events :
Join Our Newsletter :